It has been a hectic year for most of us, not just for me. Roughly two years ago we started from scratch. No money, no projects, no equipment. Now a lab has emerged. Last year field work was performed and animals were studied on and from five continents (Europe, Central America, Africa, Australia and SE Asia). I have seen more invertebrate orders and families last year than in any of the previous years. Elaborate field experiments were set up (Celina, Beth, Hendrik) sometimes with so many treatments that it was difficult not to get lost. We abandoned plankton as a core group and embraced more invertebrate and vertebrate groups than ever before. Our taxonomical expertise has increased tremendously and so has the literature we have on groups we never tackled before. Yannick and Hendrik made their own field guide for rock pool invertebrates from Western Australia, Mario personally made a key for invertebrates from moss islands in Belgium and nobody is more skilled in finding cryptic species than Gisela. I cannot tell you how much I appreciate this because this quality control and extra taxonomical resolution makes all the difference and allowed us to detect a lot of patterns that would have remained obscured otherwise.

Many of you also did exceptional things to gather data. Some people crawled through dark holes full of feces (Barbara) to get data, others will face or have faced the treacherous mud of wetlands (Evelien, Lise). Some of you have used slave labor to collect samples and aid with lab work (Celina) or seduced Greek fishermen to get free transport (Sofie). Several of you have struggled with terrible bureaucracy, permits, tropical parasites or a combination of all four. Some people said I was foolish to take on so many MSc students by the start of the year and they were right. But I was convinced by all of your plans and have not regretted it.
I’m also happy that overall we are doing well. Despite the fact that I never had less time to write papers than last year, we scored important papers in Global Ecology and Biogeography and Scientific Reports… and strangely enough in Alzheimer’s & Dementia (don’t ask me how, I forgot). Our website got more than 10 000 unique visitors.
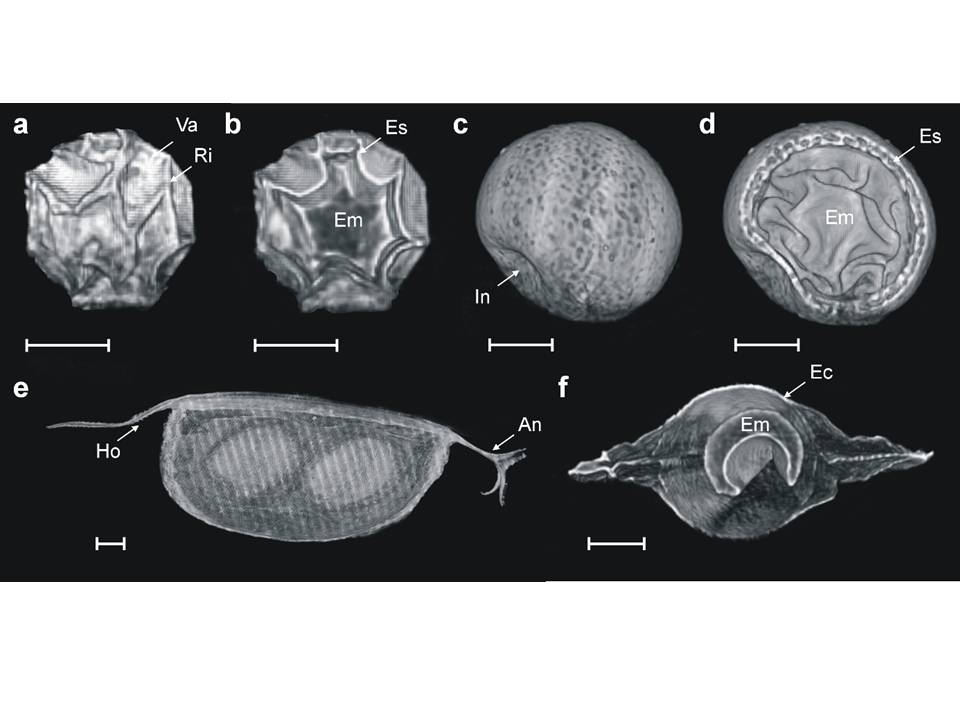
Valerio discovered something amazing in reptiles (I cannot write what, not published yet). Mathil got a PhD fellowship and lead a successful expedition into unknown territories. Evelien’s connectivity analyses are being explored in other systems and datasets from moss mites and coral reefs to pelicans. Karen found that predator avoidance strategies in the African savannah affect the shape of drinking holes and the vegetation around it… because antelope tend to approach water upwind to avoid being detected. With Melissa, we used a supercomputer to reconstruct interaction strengths in food webs. We build a matrix population model that showed that evolutionary bet hedging could help populations to cope with climate change. We joined the Bromeliad Working Group and are planning more exchange with Canada and Brazil. We used X rays to peer into the darkness inside the time capsules of dormant plankton and are only beginning to understand how they manage to use time travel to cope with environmental stochasticity. We are collaborating with Bio Engineers (the ecology of intracellular interactions), Physicists (optics), Archeologists (distribution models of ancient settlements) and Geographers (dispersal, urban ecology) on interdisciplinary research themes. These are just few of many highlights of my year.
Thanks to all my students and collaborators for helping us with starting up this lab!